Understanding Tirzepatide: A Paradigm Shift in Weight Loss Injections in New Jersey
In the evolving landscape of obesity medicine, Tirzepatide emerges as a revolutionary agent transforming safe weight loss injections in NJ. Unlike traditional weight loss methods, Tirzepatide integrates dual incretin receptor agonism, targeting both GLP-1 and GIP pathways, thereby enhancing metabolic regulation with unprecedented efficacy. This innovation presents an advanced therapeutic option for patients aiming for sustainable fat loss under physician supervision.
Mechanistic Insights into Tirzepatide’s Role in Safe Fat Reduction
Tirzepatide’s unique pharmacodynamics leverage the synergy between glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP) receptors, facilitating improved glycemic control and appetite suppression. This dual action not only optimizes energy expenditure but also modulates adipose tissue metabolism, leading to clinically significant weight reduction. Such mechanisms differentiate it from conventional GLP-1 agonists, positioning Tirzepatide as a superior injectable option within NJ’s obesity treatment protocols.
What Are the Expected Clinical Outcomes and Safety Considerations When Using Tirzepatide for Weight Loss?
Clinical trials and real-world applications in NJ have demonstrated that patients experience notable weight loss averages exceeding 15% of initial body weight over 24 to 52 weeks. However, safety profiles emphasize the importance of gradual dose escalation to mitigate gastrointestinal adverse effects such as nausea and vomiting. Physicians in NJ advocate for personalized dosing schedules and comprehensive monitoring to maximize benefits while ensuring patient safety. These protocols are essential to align with FDA-approved guidelines and optimize long-term adherence.
Integrating Tirzepatide into NJ’s Physician-Supervised Weight Loss Programs
Implementation of Tirzepatide within structured weight loss programs in NJ involves multidisciplinary coordination, emphasizing patient education, nutritional counseling, and behavioral modification alongside injection therapy. This holistic approach addresses underlying metabolic dysfunctions and supports sustained fat loss. Experienced NJ clinicians tailor injection plans to individual metabolic profiles, enhancing efficacy and reducing risks. For detailed program options, patients can explore customized Tirzepatide injection plans that exemplify best practices in safe weight loss injections.
Expertise-Driven Patient Selection: Who Benefits Most from Tirzepatide Injections in NJ?
The optimal candidates for Tirzepatide are adults with obesity (BMI ≥30) or overweight individuals (BMI ≥27) with comorbidities such as type 2 diabetes or cardiovascular risks. NJ specialists emphasize a thorough evaluation of patient history, metabolic markers, and prior weight loss attempts to ensure the appropriateness of this injectable therapy. This nuanced selection process enhances therapeutic outcomes and minimizes contraindications, aligning with evidence-based obesity medicine standards.
Encouraging Professional Dialogue and Knowledge Sharing
Healthcare providers and patients interested in the latest advancements in safe weight loss injections with Tirzepatide are encouraged to engage with NJ’s expert resources. For comprehensive insights and updates, visit NJ’s Expert Guide to Safe Tirzepatide Injections. Contributing your professional experiences enriches the community’s collective expertise and promotes optimized patient care.
For authoritative research on Tirzepatide’s efficacy and safety profile, see the New England Journal of Medicine’s SURPASS clinical trial publication, which provides robust data supporting its use in weight management.
Addressing Common Myths and Misconceptions About Tirzepatide in NJ Clinics
Despite Tirzepatide’s growing recognition as a transformative therapy in obesity medicine, several misconceptions persist among patients and some healthcare providers. One prevalent myth is that Tirzepatide functions merely as an appetite suppressant. In reality, its dual GLP-1 and GIP receptor agonism offers multifaceted benefits, including enhancement of insulin sensitivity, modulation of lipid metabolism, and improvement of energy expenditure. This comprehensive metabolic impact distinguishes Tirzepatide from traditional weight loss medications, underscoring the importance of patient education to set accurate expectations.
Another concern often voiced pertains to the safety profile, particularly regarding gastrointestinal side effects and long-term use. NJ physicians emphasize that gradual dose titration and close monitoring within physician-supervised programs significantly reduce adverse events, ensuring patient adherence and safety. Additionally, emerging data suggest potential cardiovascular benefits, which are especially relevant for patients with obesity-related comorbidities.
Innovative Protocols: How NJ Physicians Personalize Tirzepatide Dosing for Optimal Outcomes
Personalization of Tirzepatide therapy is a hallmark of effective weight loss programs in New Jersey. Clinicians incorporate patient-specific variables such as baseline metabolic rate, coexisting health conditions, and previous pharmacologic responses to tailor dosing regimens. Initiating treatment at lower doses with incremental adjustments allows for minimization of side effects and maximization of fat loss efficacy. This approach reflects a precision medicine model within obesity care, facilitating sustainable results and improved quality of life.
Can Combining Tirzepatide with Lifestyle Interventions Unlock Superior Weight Loss Results?
Integrating Tirzepatide therapy with structured lifestyle modifications—including tailored nutrition plans, physical activity, and behavioral counseling—can amplify weight loss outcomes. NJ experts advocate for this integrative strategy, as pharmacotherapy alone may not address behavioral and environmental factors contributing to obesity. Ongoing clinical observations support the synergistic effect of combining Tirzepatide with comprehensive lifestyle interventions, leading to enhanced metabolic improvements and patient empowerment.
For patients interested in embarking on this multifaceted journey, exploring programs like the Tirzepatide weight loss programs in NJ offers access to personalized care plans developed by experienced obesity medicine specialists.
Evaluating the Broader Impact: Tirzepatide’s Role in Addressing New Jersey’s Obesity Epidemic
With obesity rates remaining a public health challenge in New Jersey, Tirzepatide represents a promising tool to mitigate this epidemic through physician-guided interventions. Its efficacy in promoting substantial and sustained fat loss can reduce the prevalence of obesity-associated complications, such as type 2 diabetes and cardiovascular disease. NJ healthcare systems are increasingly integrating Tirzepatide into multidisciplinary obesity treatment frameworks, fostering population health improvements.
According to a 2023 review by the National Center for Biotechnology Information (NCBI), Tirzepatide’s dual incretin receptor targeting not only supports weight loss but also offers metabolic benefits that may transform obesity management on a systemic level. This underscores the medication’s significance beyond individual patient outcomes to broader healthcare implications.
Healthcare providers and patients alike are encouraged to engage with local expert resources to stay abreast of evolving evidence and treatment innovations. Sharing experiences and outcomes through forums and consultations enriches NJ’s collective expertise, enhancing care quality.
Have you experienced or prescribed Tirzepatide as part of a physician-supervised weight loss program? Share your insights and questions in the comments to foster professional dialogue and community learning.
Precision Medicine in Tirzepatide Therapy: Customizing Dosing Beyond Standard Protocols
While FDA-approved dosing guidelines for Tirzepatide provide a foundational framework, leading obesity specialists in New Jersey increasingly adopt a precision medicine approach to optimize patient outcomes. This involves integrating advanced biomarker analysis such as insulin resistance indices, adipokine profiles, and even genetic polymorphisms affecting incretin receptor sensitivity. By tailoring dose escalation schedules and maintenance doses to these individualized metabolic signatures, clinicians can enhance efficacy while minimizing adverse effects, particularly gastrointestinal discomfort.
For example, patients exhibiting heightened sensitivity to GLP-1 receptor agonism may benefit from slower titration schedules, while those with suboptimal glycemic control might require more aggressive dose adjustments combined with adjunctive therapies. This meticulous customization underscores the transition from a one-size-fits-all model towards a nuanced, patient-centric paradigm within New Jersey’s weight loss injection programs.
Integrative Metabolic Modulation: Combining Tirzepatide with Emerging Adjunctive Therapies
Beyond lifestyle interventions, research in NJ obesity clinics is exploring the synergy of Tirzepatide with novel adjunctive treatments such as time-restricted feeding, gut microbiome modulation, and targeted nutrient supplementation. These integrative strategies aim to potentiate Tirzepatide’s metabolic effects by addressing complementary physiological pathways involved in energy homeostasis and adipose tissue remodeling.
Preliminary clinical observations suggest that coupling Tirzepatide with prebiotic-rich diets or probiotic therapies may enhance gut hormone secretion and improve metabolic flexibility. Similarly, integrating intermittent fasting protocols tailored to patient circadian rhythms can amplify fat oxidation and optimize the hormonal milieu, further supporting sustainable weight loss.
How Does Tirzepatide Influence Brown Adipose Tissue Activation and Energy Expenditure?
Emerging evidence indicates that Tirzepatide’s dual agonism extends beyond appetite suppression and glycemic control to affect adipose tissue phenotypes. Specifically, Tirzepatide may stimulate activation of brown adipose tissue (BAT), a metabolically active fat depot responsible for thermogenesis and increased energy expenditure. This BAT activation contributes to enhanced basal metabolic rate and improved lipid metabolism, aiding in more effective fat loss.
Research published in Nature Medicine illustrates that GLP-1 and GIP receptor co-activation promotes browning of white adipose tissue, thus potentiating metabolic benefits beyond traditional weight loss mechanisms. This insight is pivotal for NJ clinicians aiming to harness Tirzepatide’s full therapeutic potential, as it informs adjunctive treatments and patient counseling on expected physiological changes.
Understanding these complex metabolic interactions empowers providers to design comprehensive interventions that transcend mere calorie restriction, embracing a systems biology perspective essential for durable obesity management.
To deepen your understanding and explore personalized treatment options incorporating these advanced protocols, consider consulting with NJ’s top obesity medicine specialists who specialize in Tirzepatide therapy integration.
Refining Tirzepatide Therapy Through Molecular and Genetic Profiling
Contemporary obesity treatment in New Jersey is rapidly embracing precision medicine paradigms to optimize Tirzepatide administration. By incorporating comprehensive molecular and genetic profiling, clinicians identify patient-specific biomarkers such as polymorphisms in the GLP-1 and GIP receptor genes, variations in adipokine secretion, and insulin sensitivity indices. These insights inform individualized titration schedules and adjunct therapies, allowing for maximized efficacy while mitigating adverse gastrointestinal events commonly seen during dose escalation.
This granular approach transcends conventional dosing algorithms, enabling a dynamic modulation of therapy tailored to each patient’s unique metabolic landscape. The integration of such biomarker-driven strategies is setting new benchmarks for patient outcomes in obesity pharmacotherapy within NJ clinical settings.
Synergistic Metabolic Modulation: Tirzepatide in Concert with Microbiome and Chrononutrition Interventions
Innovative research spearheaded by NJ obesity centers is elucidating the potentiation of Tirzepatide’s effects through adjunctive interventions targeting the gut microbiome and circadian biology. Time-restricted feeding protocols synchronized with patients’ circadian rhythms have demonstrated enhanced mitochondrial efficiency and fat oxidation when combined with Tirzepatide therapy. Concurrently, modulation of gut microbial populations via prebiotic and probiotic supplementation appears to augment endogenous incretin hormone secretion, thereby reinforcing Tirzepatide’s dual agonism mechanisms.
Such integrative metabolic strategies represent a paradigm shift, positioning Tirzepatide not merely as a pharmacologic agent but as a central component of a multifactorial treatment architecture addressing systemic energy homeostasis and adipose tissue plasticity.
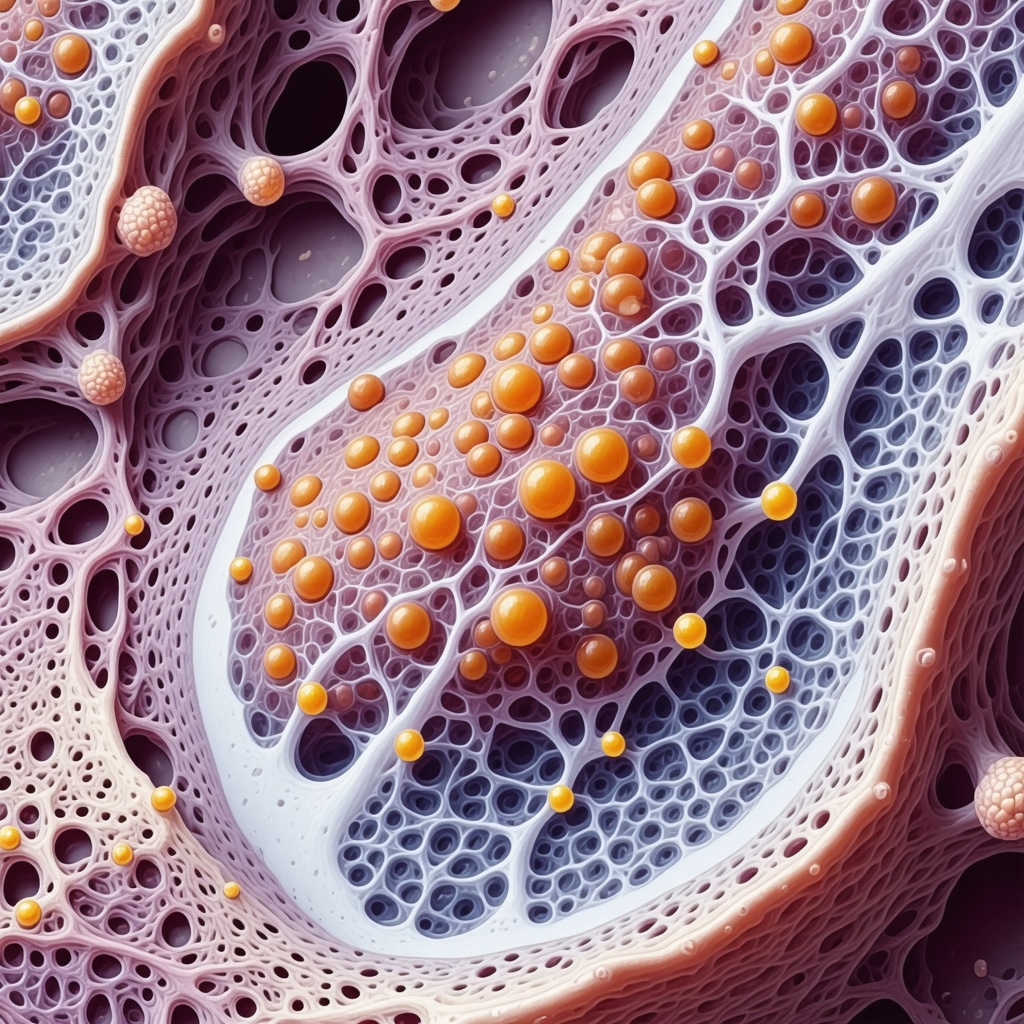
How Does Tirzepatide Influence Brown Adipose Tissue Activation and Energy Expenditure?
Emerging evidence indicates Tirzepatide’s dual receptor agonism extends to promoting brown adipose tissue (BAT) activation, a process pivotal for thermogenesis and heightened basal metabolic rate. Activation of BAT leads to increased energy expenditure independent of physical activity, a critical factor in sustainable weight management. Furthermore, co-activation of GLP-1 and GIP receptors induces browning of white adipose tissue, as demonstrated in recent studies published in Nature Medicine. This metabolic remodeling facilitates improved lipid utilization and insulin sensitivity, enhancing the overall therapeutic profile of Tirzepatide beyond appetite suppression.
NJ clinicians leveraging this knowledge can incorporate targeted lifestyle and nutritional recommendations to potentiate these adipose tissue transformations, fostering more durable fat loss outcomes.
Extending Frontiers: Combining Tirzepatide with Emerging Adjuncts in Clinical Practice
Ongoing clinical investigations in New Jersey explore the synergistic potential of Tirzepatide with novel adjunctive therapies such as intermittent fasting, circadian-aligned meal timing, and nutraceuticals designed to modulate adipocyte function. These combinations aim to amplify mitochondrial biogenesis and enhance metabolic flexibility, addressing obesity’s multifactorial etiology.
Such advanced therapeutic frameworks underscore the necessity for continuous professional education and interdisciplinary collaboration to harness these cutting-edge interventions effectively.
For comprehensive guidance and personalized program integration, NJ healthcare professionals and patients are encouraged to consult with specialized obesity medicine experts proficient in these sophisticated multimodal strategies.
Explore the forefront of Tirzepatide therapy by connecting with NJ’s leading specialists and transform your approach to physician-supervised weight loss injections.
Expert Insights & Advanced Considerations
Precision Medicine Elevates Tirzepatide Efficacy
Leveraging molecular and genetic profiling to tailor Tirzepatide dosing transcends standard protocols, enabling clinicians in New Jersey to optimize therapeutic outcomes while minimizing adverse effects. This approach integrates biomarkers such as incretin receptor polymorphisms and insulin sensitivity indices, fostering truly individualized obesity treatment plans.
Synergistic Metabolic Modulation Enhances Fat Loss
Combining Tirzepatide with adjunctive interventions—like time-restricted feeding, circadian-aligned nutrition, and microbiome-targeted therapies—creates a multifaceted metabolic environment. This synergy potentiates mitochondrial biogenesis, brown adipose tissue activation, and hormonal balance, thereby amplifying sustainable weight loss beyond pharmacology alone.
Brown Adipose Tissue Activation as a Therapeutic Mechanism
Emerging data reveal Tirzepatide’s role in stimulating brown and beige adipose tissue thermogenesis, which increases basal metabolic rate independent of physical activity. This mechanism expands the understanding of how dual GLP-1 and GIP receptor agonism contributes to durable fat loss, offering NJ clinicians actionable insights for adjunct lifestyle counseling.
Personalized Titration Minimizes Gastrointestinal Side Effects
Gradual dose escalation tailored to patient-specific metabolic sensitivity reduces common gastrointestinal adverse events, improving adherence and long-term success. NJ practitioners emphasize this careful titration within physician-supervised programs to maintain safety and maximize fat loss efficacy.
Integrating Tirzepatide Into Multidisciplinary Obesity Care
Effective Tirzepatide therapy in New Jersey is embedded within comprehensive weight loss programs that combine pharmacotherapy with behavioral, nutritional, and psychological support. This holistic framework addresses obesity’s multifactorial nature, enhancing overall patient outcomes and quality of life.
Curated Expert Resources
New England Journal of Medicine – SURPASS Clinical Trial: Provides pivotal, peer-reviewed evidence on Tirzepatide’s efficacy and safety profile, essential for understanding clinical trial benchmarks.
National Center for Biotechnology Information (NCBI) Reviews: Offers comprehensive analyses of Tirzepatide’s metabolic mechanisms and emerging research trends, supporting advanced clinical decision-making.
Nature Medicine Publications on Adipose Tissue Biology: These studies elucidate the molecular underpinnings of brown and beige fat activation relevant to Tirzepatide’s therapeutic effects.
NJ Expert Guide to Safe Tirzepatide Injections (weightlossdoctornj.com): A localized, authoritative resource integrating best practices for Tirzepatide administration within NJ’s clinical context.
WeightLossDoctorNJ Tirzepatide Program Resources: Customized plans and patient-centric protocols that exemplify physician-supervised care excellence in New Jersey.
Final Expert Perspective
Tirzepatide is undeniably reshaping the landscape of physician-supervised weight loss injections in New Jersey by introducing a sophisticated, mechanism-driven approach to obesity management. Its dual incretin receptor agonism not only facilitates robust fat loss but also engages complex metabolic pathways—including brown adipose tissue activation and microbiome interactions—that extend benefits beyond traditional therapies. The integration of precision medicine and interdisciplinary care further amplifies patient outcomes, setting a new standard for safe and effective weight loss interventions.
For clinicians and patients committed to advancing obesity treatment, engaging with state-of-the-art Tirzepatide protocols and expert resources is imperative. Explore comprehensive guidance through the NJ Expert Guide to Safe Tirzepatide Injections and consider personalized consultations to tailor your approach. Your insights and experiences are invaluable—join the conversation with leading New Jersey specialists to elevate care quality and patient success.